 electron geometry = octahedral molecular geometry = octahedral S :F : : : : : :F: F: F: : :F :F: : : : : Predicting Molecular Geometry Example 11: SF6 AX6E0 Six electron pairs around the central atom. 90° Square planar Square pyramid Octahedral Octahedral (Electron Geometry)Six e- pairs about central atom Equivalent atoms electron geometry = trigonal bipyramidal molecular geometry = linear : : F : : Xe : : : F : : Predicting Molecular Geometry Example 10: XeF2 AX5E0 AX2E3 Five electron pairs around the central atom. 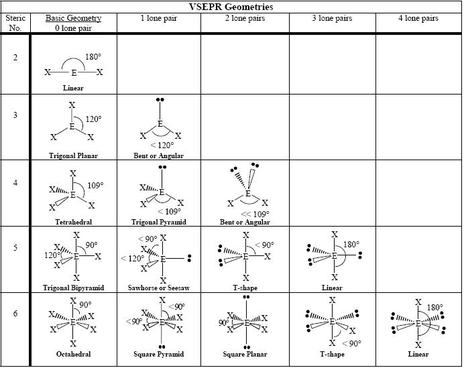
electron geometry = trigonal bipyramidal molecular geometry = T-shaped : : : F : : : F Br : : : : F : Predicting Molecular Geometry Example 9: BrF3 AX5E0 AX3E2 Five electron pairs around the central atom. electron geometry = trigonal bipyramidal molecular geometry = seesaw : : F : : F : : S : : F : : : F : Predicting Molecular Geometry Example 8: SF4 AX5E0 AX4E1 Five electron pairs around the central atom. electron and molecular geometry= trigonal bipyramidal : : : F : F : : F : : : : : F : P : F : : Predicting Molecular Geometry Example 7: PF5 AX5E0 Five electron pairs around the central atom. **Put lone pairs in the equatorial positions. Green atoms are axial blue atoms are equatorial. 120° Seesaw T-shaped Linear Triangular bipyramidal Trigonal Bipyramidal (Electron Geometry)Five e- pairs about central atom The atoms are non-equivalent. Predicting Molecular Geometry Tetrahedral - bond angles Order of increasing repulsion: bonding pair-bonding pair < bonding pair-lone pair < lone pair-lone pairĩ0°. O H H electron geometry = tetrahedral molecular geometry = angular or bent H O H Predicting Molecular Geometry Example 6: AX4E0 AX2E2 H2O Four electron pairs around the central atom. molecular geometry = triangular pyramidal H N H H N H H H Predicting Molecular Geometry Example 5: AX4E0 AX3E1 NH3 Four electron pairs around the central atom. tetrahedral electron pair geometry = molecular geometry H C H H H Predicting Molecular Geometry Example 4: AX4E0 CH4 Four electron pairs around the central atom. Tetrahedral (Electron Geometry)Four e- pairs about central atom bond lone pairs pairs 4 0 tetrahedral Model 3 1 triangular pyramidal 2 2 angular (bent) O S O S O O Predicting Molecular Geometry Example 3: SO2 AX3E0AX2E1 Three electron pairs around the central atom. AX3E0 Three electron pairs around the central atom. Predicting Molecular Geometry Example 2: BF3. Trigonal Planar (Electron Geometry)Three e- pairs about central atom bond lone Molecular pairs pairs Geometry Model 3 0 triangular planar 2 1 angular (bent) 1 2 linear
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |



 RSS Feed
RSS Feed
